FeedingGuilds
Feeding Guilds and Trophic Levels
Feeding Guilds
As Merriam-Webster dictionary defines it, a feeding guild is “a group of organisms that use the same ecological resource in a similar way."[1] In simpler terms, feeding guilds can be described as groups of living things that get their energy necessary to survive from the same source. Feeding guilds do not have to be made up of the same or similar species; they can be completely unrelated, only alike in the fact that they acquire resources from their surroundings in similar ways. Because members of a guild use the same resources, competition is common within that guild.[2] A feeding guild is composed of three major categories: autotrophs, heterotrophs, and detritivores.
Autotrophs
Autotrophs are "self-nourishing organisms capable of absorbing solar energy and photosynthetically building high-energy organic substances." [3] Because these organisms need light to survive, they are most commonly found within the top 70 meters of water in marine environments. Since most coral reefs reside in shallower waters, they provide a great environment for autotrophs. Two examples of autotrophs in coral reef ecosystems are seaweed and zooxanthellae, a type of algae that makes up part of the coral and gives it color.[4]
Heterotrophs
Heterotrophs are consumers, they obtain their energy necessary to survive from other organisms. Heterotrophs can be broken down into seven different subcategories: herbivores, carnivores, omnivores, filter feeders, planktivores, oppurtunistic feeders, and parasites.
Herbivores
Herbivores feed only upon autotrophs. There is a large guild of herbivores in coral reef environments.[4] Two examples are the Green Sea Turtle, which eats sea grass, and a sea urchin, which eats algae.
Carnivores
Carnivores feed only upon other animals. In coral reefs, they range from top predators such as barracudas and eels to smaller predators such as lobsters.
Omnivores
Omnivores consume both autotrophs and other heterotrophs.An example of an omnivore in coral reefs are angelfish which feed algae, smaller fish, and shrimp.
Planktivores
Planktivores eat the small organisms (collectively known as Plankton) that drift in the water. They are herbivorous and consume primarily phytoplankton, or they can be zooplanktivores that consume suspended animals. [7] The whale shark, which feeds primarily on plankton, is an example of planktivores in coral reef ecosystems.
Filter/suspension feeding are the primary methods of consuming this organic material (pseudo-plankton) that floats in the water column.[7] Suspension feeders actively capture particles of food from the water using tentacles while filter feeders use a filtration system to filter out dissolved particles after pumping in water. Zooplankton and sponges are good examples of these in coral reef environments.[8]
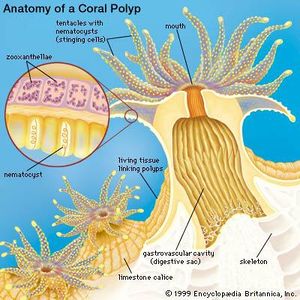
One of the most well-known examples of filter feeders are corals. Corals use suspension feeding to supplement the nutrients they already derive from their symbiotic relationship with Zooxanthellae, and they tend to use it only during the night[9].When suspension feeding, the coral polyp extends its tentacles out from its body where they can encounter small fish, zooplankton, bacterioplankton, and other such plankton. The surface of each tentacle has thousands of stinging cells known as nematocysts, which are fired when prey swims or floats by. The stinging action of the nematocysts stuns or kills the prey and then the polyp tentacles pass the prey item back into the mouth of the polyp[9]. Some species of coral are mucus suspension feeders, which means that they use mucus films or strands to collect fine particles from the water column rather than nematocysts. Cilia are then used to draw the trapped particles into the polyp’s mouth[9][10].
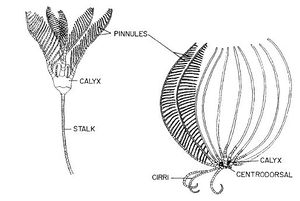
Crinoids are another example of an organism that uses suspension feeding. Crinoids have finger-like podia that occur in triads on both sides of the pinnular ambulacra. When a suspended food particle touches the podia, it flicks, bends, or curls rapidly inward to force the food particle into its food groove. The primary podia are adhesive, but the role of mucus in capturing food particles varies among species. [11].
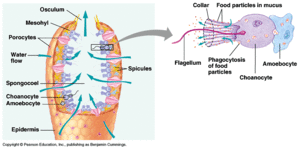
Sponges (Porifera) are prime examples of filter feeders. Sponges filter feed by drawing water in through incurrent pores. They draw this water in by creating a current with special collar cells that have whip-like flagella.[10]. As water passes through the channels and chambers inside of the sponge, particles are filtered out and taken up by the sponge as food[13]. The filtered water is then pumped out through a large opening at the top of the sponge called the osculum[10]. Sponges are unique among filter feeders in that they are the only filter-feeding metazoan group capable of capturing and consuming picoplankton as small as 0.1 micrometers. They are able to do this thanks to a very fine internal mesh network used to collect the tiny particles from the water [14].
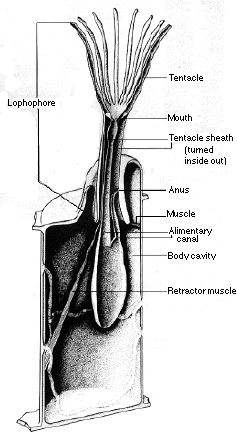
Bryozoans also are filter feeders. They feed on plankton using a ring of tentacles called the Iophophore. The tentacles on this ring are ciliated and are retracted through contractile muscles that are activated by nerve impulses [10].
Brachiopods are very similar to Bryozoans in that they also have a ciliated Iophophore used for filter feeding. The unique feature of Brachiopods is that they have two coiled tentacles within their shell that form the Iophophore [10].
Polychaete worms belonging to the family Serpulidae and to family Sabellidae are filter feeders. Well-known examples include Featherduster Worms and Christmas Tree Worms, which are both members of the Serpulidae family. They extend tentacles out to feed and then use cilia to sweep water towards the mouth and to filter out food particles.[10]
One final example of filter feeders are the tunicates. Tunicates draw in hundreds of liters of water a day through an incurrent siphon. This water passes through a pharynx where small food particles are filtered out before the water is expelled via the excurrent siphon. These small food particles are trapped in a continually moving layer of mucus produced by specialized cells. Cilia are responsible for both creating a current of water flow within the tunicate and for moving mucus and food along to the digestive system[16].
Fishes - see Other Techniques Page
Opportunistic feeders
This group targets prey based on what is available at the time. Scavengers and cannibals, such as some species of crabs, are included in this group. [7]
Parasites
Parasites "live on or in another organism", taking their nutrition from the host organism while giving nothing in return. [7] A common example seen on coral reef fish is the cymothoid isopod which can deplete energy and even leave bone deformations to the affected fish's facial area.[17]
Detritivores

Detritovores are decomposers that feed upon the waste products and dead remains of other organisms. [3] They are extremely important to coral reef ecosystems because the break down the dead waste, convert it to energy they can use, and then return important materials back into the environment. For coral reefs, the main decomposers are bacteria. Bacteria play an essential role in the nitrogen cycle to provide the environment with necessary nutrients, such as nitrate, while also keeping toxic waste levels low.[4]
Bacteria are not the only detritivores on the reef, however. For example, sea cucumbers constantly funnel sand and mud into mouth to extract organic material. By doing this, they effectively clean reef of any dead organic matter and any bacteria that may be living on it. In fact, it is estimated that detritus-eating sea cucumbers on typical coral reef will cycle all the surface sediment surrounding that reef through their digestive tracts at least twice a year[10].
Many species of fish are also considered detritivores - see Other Feeding Techniques
Trophic Levels
Trophic levels describe the transfer of chemical energy and nutrients through the food chain.[19] Each trophic level represents organisms who serve the same role in the food chain. The trophic levels that will be discussed are the primary prodcuers, primary consumers, secondary consumer, tertiary consumers, and decomposers. The first trophic level begins at the bottom of the food chain where food energy is initially synthesized. Then, each subsequent trophic level represents the group of organisms which feed on the previous trophic level, or the next level in the food chain. As energy moves up a trophic level, only some percentage is converted into usable energy. The rest is lost through heat or other forms of energy. This means that only a smaller fraction of the initial energy produced remains at the highest trophic level. This trend with energy is also seen in biomass. The highest trophic level has the smallest biomass, and the lowest trophic level has the largest biomass. It is possible to represent this relationship graphically, and the simplest model is the trophic pyramid shown below.[19]
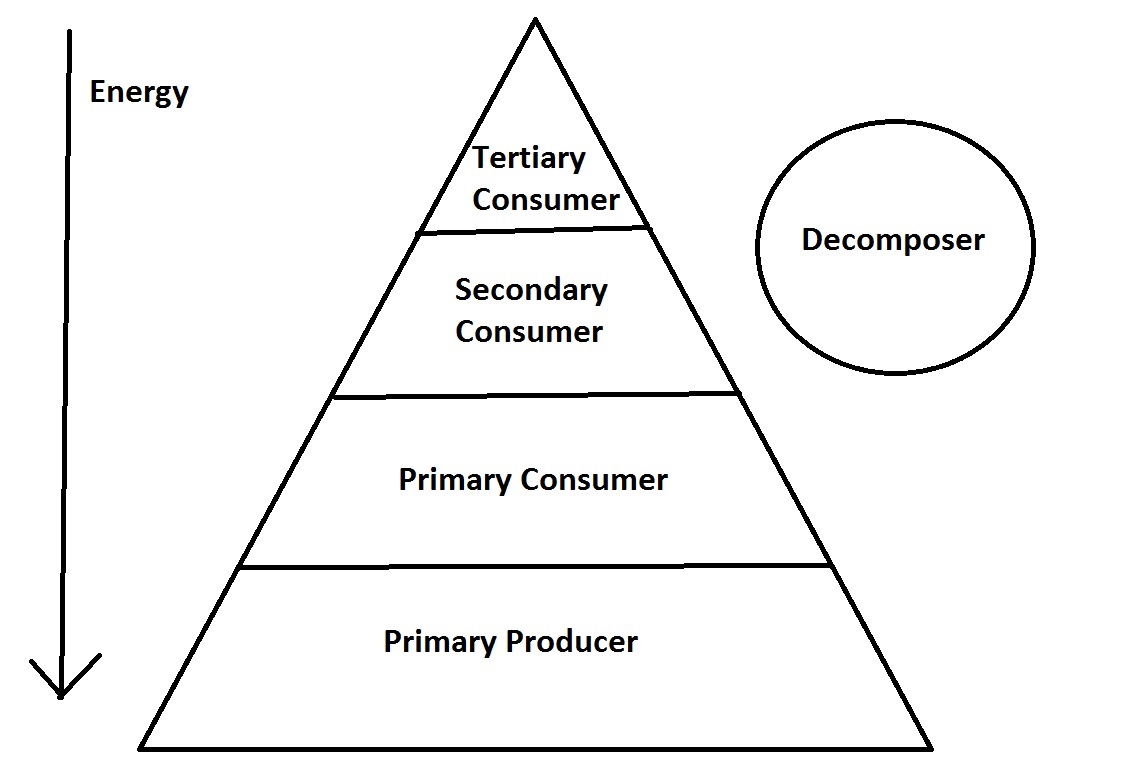
The trophic levels form this pyramid shape to represent the energy (biomass) lost through each trophic level. This is a highly simplified model. There could be a greater number of trophic levels or overlap between levels. A more complex and realistic model would be an interconnected food web. [19]
Primary Producer
Primary producer are the lowest trophic level and the lowest organisms on the food chain. These are the organisms that produce their own chemical energy through photosynthesis. Autotrophs are primary producers.[19]
Primary Consumer
Primary consumers consume primary producers. Heterotrophic organisms that are typically fall in the primary consumer tropic level are herbivores, filter/suspension feeders, and planktivores.[19]
Secondary Consumer
Secondary Consumers feed on primary consumers. Heterotrophic organisms that fall into the secondary consumer tropic level are lower level carnivores and possibly omnivores.[19]
Tertiary Consumer
Tertiary consumers are the top of the food chain and do not have an predators. They feed on secondary consumers. Heterotrophic organisms that fall into the tertiary consumer tropic level are higher level carnivores.[19]
Decomposer
Decomposers feed on dead organic material from any trophic level in the food chain. For this reason, decomposer do not hold a position in the energy pyramid, and are instead located on the side of the pyramid. Detritivores and scavengers are decomposers.[19]
Why It's Important: Imbalances in the System
“Coral reefs are complex ecosystems that require a balanced trophic structure to function properly and efficiently. Imbalances can occur in this intricate trophic cascade from the top down or the bottom up.”[4]
An example of bottom up effect imbalances that illustrates why feeding guilds and trophic levels are important to the coral reef ecosystem comes from an incident in the Great Barrier Reefs. Coral in the Great Barrier Reefs is now being threatened not only by coral bleaching but an even bigger predator, the Crown of thorns starfish. Agricultural runoff containing fertilizer has caused an excess of phytoplankton to be present in the waters around the reefs. The Crown of thorn starfish larvae feed on this phytoplankton and thus caused a population explosion in the area. The problem is that the crown of thorn starfish feeds on coral and can eat up to 10 square meters of tissue a year. This bottom up effect has taken its toll on the reefs surrounding Australia, with 42% of coral reef decline in the area due to the crown of thorns invasion. [20]
References
- ↑ "Guild." Merriam-Webster. Merriam-Webster, n.d. Web. 15 Apr. 2014. <http://www.merriam-webster.com/dictionary/guild>
- ↑ Thompson, John. "Guild (ecology)." Encyclopedia Britannica Online. Encyclopedia Britannica, n.d. Web. 15 Apr. 2014. http://www.britannica.com/EBchecked/topic/248611/guild
- ↑ 3.0 3.1 Sumich, James L. An Introduction to the Biology of Marine Life, Seventh Edition. WCB/McGraw Hill. 1999.
- ↑ 4.0 4.1 4.2 4.3 "Coral reef ecology." coralscience.org. N.p., n.d. Web. 15 Apr. 2014. http://www.coralscience.org/main/articles/climate-a-ecology-16/coral-reef-ecology
- ↑ "Coral Reefs." Geo for CXC. 2014. Accessed April 19, 2015. http://www.geoforcxc.com/water/coral-reefs/.
- ↑ Horsfall, Sophie. "Cenolia Glebosis: Black Featherstar." Great Barrier Reef Invertebrates. 2014. Accessed April 19, 2015. http://www.gbri.org.au/SpeciesList/Cenoliaglebosus%7CSophieHorsfall.aspx?aid=48&PageContentID=5263.
- ↑ 7.0 7.1 7.2 7.3 Tackett, Denise and Larry. Reef Life: Natural History and Behaviors of Marine Fishes and Invertebrates. Neptune City, NJ: T.F.H Publications, 2002, p. 117-118. Print.
- ↑ "Hentschel, B.T. "Suspension Feeders." Ecology Center. May 16, 2013. Accessed April 15, 2015. http://www.ecologycenter.us/population-dynamics-2/suspension-feeders.html.
- ↑ 9.0 9.1 9.2 "What Are Coral Reefs." CoRIS. January 1, 2015. Accessed April 15, 2015. http://www.coris.noaa.gov/about/what_are/.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 Strykowski, Joe. & Bonem, Rena Mae,. (1993). Palaces under the sea : a guide to understanding the coral reef environment. Crystal River, FL : Star Thrower Foundation.
- ↑ "The Crinoid Feeding Mechanism." Nova Southeastern University. January 1, 1997. Accessed April 15, 2015. http://www.nova.edu/ocean/messing/crinoids/8 Feeding mechanism.html.
- ↑ "Sponges." Accessed April 19, 2015. http://facultyweb.brynmawrschool.org/UpperSchool/Science/Classes/AllBiology/bio/biotext/biowilson/2009-2010/HONORS/EOL Honors1/Fallon/Site/Sponges.html.
- ↑ "Filter-Feeding in Reef Sponges." NOAA. January 1, 2008. Accessed April 15, 2015. http://coralreef.noaa.gov/education/educators/resourcecd/lessonplans/resources/sponge_lp.pdf
- ↑ Reiswig, H.M.. 1975. BACTERIA AS FOOD FOR TEMPERATE-WATER MARINE SPONGES. Canadian Journal of Zoology-Revue Canadienne De Zoologie 53: 582-589.
- ↑ "Bryozoans." Paleontological Laboratory at SUNY Cortland. Accessed April 19, 2015. http://paleo.cortland.edu/tutorial/Bryozoans/bryozoans.htm.
- ↑ "Tunicates." Marine Education Society of Australia. January 1, 2015. Accessed April 15, 2015. http://www.mesa.edu.au/tunicates/.
- ↑ Sullivan, M., and R. Stimmelmayr. "Cymothoid isopods on coral reef fishes in the near shore marine environment of St. Kitts, Lesser Antilles." Proceedings of the 11 th International Coral Reef Symposium 26 (2008): 1394-1395. Print.
- ↑ Grall, George. "Donkey Dung Sea Cucumber Foraging for Detritus on a Sandy Sea Floor." AllPosters. Accessed April 19, 2015. http://www.allposters.co.uk/-sp/Donkey-Dung-Sea-Cucumber-Foraging-for-Detritus-on-a-Sandy-Sea-Floor-Posters_i6111993_.htm.
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 19.7 Reece, Jane; Taylor, Martha; Simon, Eric; Dickey, Jean. Campbell Biology Concepts and Connections. San Fransico, CA: Pearson Education, Inc., 2012, p. 744-745. Print.
- ↑ Griffiths, Sarah. "Invasion of carnivorous starfish could be a greater threat to the Great Barrier Reef than global warming, experts claim." Mail Online. Associated Newspapers, 17 Feb. 2014. Web. 15 Apr. 2014. http://www.dailymail.co.uk/sciencetech/article-2561462/Invasion-carnivorous-starfish-greater-threat-Great-Barrier-Reef-global-warming-experts-claim.html