SCTLD
Stony Coral Tissue Loss Disease (SCTLD)
Stony Coral Tissue Loss Disease (SCTLD), discovered in 2014, is a contagious waterborne disease killing stony corals in the Caribbean off Jamaica, Mexico, Saint Maarten, the US Virgin Islands, Dominican Republic, Turks & Caicos Islands, Saint-Martin, Belize, Sint Eustatius, The Bahamas, Puerto Rico, British Virgin Islands, Cayman Islands, Guadeloupe, St. Lucia, Honduras and Martinique. Sick colonies display multifocal lytic necrosis, appearing as lesions, that start in the gastrodermis and extend out to the surface epithelia. Highly susceptible species are the meandroid corals–i.e., pillar corals (Dendrogyra cylindrus), elliptical star corals (Dichocoenia stokesii), smooth flower corals (Eusmilia fastigiata) and maze corals (Meandrina spp.). Starlet corals that develop numerous "blotchy" lesions, as well as diverse brain and star (boulder) corals, are also dying fairly quickly, followed by star corals (Orbicella spp., Montastraea cavernosa) and other coral species.[1]
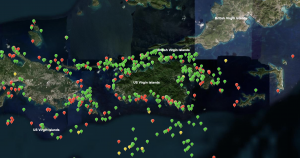
One important resource to improve public outreach is the Tracking Map created by the Atlantic and Gulf Rapid Reef Assessment organization.[2] This interactive map displays location based reef assessments of SCTLD by combining numerous coral surveys throughout the world.
There is limited understanding of SCTLD disease, including its cause. Researchers can confirm that it is waterborne, but are unsure whether it is bacterial, viral, or environmental. The lack of research makes treatment rather difficult, but here are a few ways in which scientists are trying to help coral communities.
Genetic Expression to Prevent SCTLD
A 2021 study by the University of Miami (UM) Rosenstiel School of Marine and Atmospheric Science and in collaboration with the Mote Marine Laboratory and the Smithsonian Marine Station has been the first of its kind to document coral gene response against SCTLD. Their work explores the genetic immune response of infected O. faveolata and M. cavernosa corals by exposing these healthy corals to samples of infected corals collected from reefs. The intent of this research is to document the corals' immune responses in order to understand how SCTLD affects the corals' biological mechanisms and to compare species to interpret varied responses and their outcomes.
Experiments between 2019 and 2020 concluded that SCTLD is evoking the feedback of a network of genes important for cell responses including cell death, immunity and tissue rearrangement, indicating that the disease is causing swift cell death and rearrangement of the tissue.[3] Overall, diseased O. faveolata had 2194 differentially expressed genes (DEGs) compared with healthy colonies, whereas diseased M. cavernosa had 582 DEGs compared with healthy colonies.[1]

This research is important to understand the symptoms of the disease throughout the coral body in order to diagnose and treat as soon as possible. The goal is to treat coral disease like human medicine and with further research, scientists can create a toolkit to better diagnose corals and better inform policies to help save them.[4] This analysis has been the first step toward genetically understanding SCTLD and opens doors into further research. Scientists from this study propose that future research examines early time points of infection, before the presence of lesions, to understand the activating mechanisms involved in SCTLD.[1]
Topical amoxicillin treatment for SCTLD
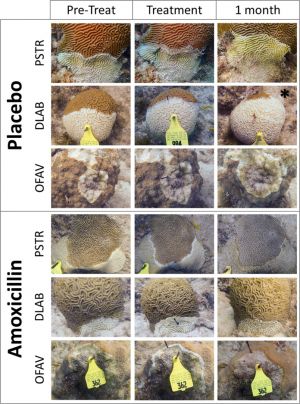
Only a handful of studies have used antibiotics to treat diseased coral. Currently, one of the prime methods of SCTLD treatment is topical amoxicillin, which is commonly used to treat bacterial infections in humans and animals. Before SCTLD interventions included topical antibiotics, it was found that antibiotic-treated water mitigates SCTLD transmission within coral colonies in lab settings.[6][7] Shortly thereafter, methods to apply topical amoxicillin to wild corals infected with SCTLD were developed, and now over 20 coral species in the Caribbean are undergoing amoxicillin treatments.[5][8]
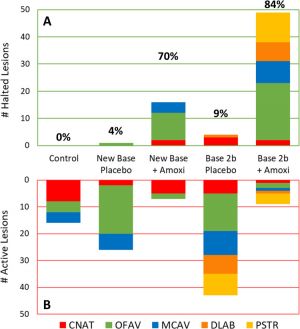
Topical amoxicillin treatments have been found to be highly effective at slowing the spread of SCTLD and in healing SCTLD lesions, but they are not capable of completely stopping its spread. Treated colonies must be re-visited after divers apply the first treatment to see if more treatment is necessary. This varies between species as some are more susceptible to SCTLD. If antibiotics fail, the next step is often amputation of the lesion.[9] Despite its effectiveness in stemming the spread of SCTLD lesions, there are many barriers to effective amoxicillin treatments. Treatments were found to be the least effective in brain corals, possibly because their deep grooves make application more difficult. Also, topical treatment benefits mostly individual corals receiving the treatment and rarely the entire colony, which makes effective applications labor-intensive and expensive.[5] Additionally, offshore sites are more prone to reinfection than inshore sites after being treated with amoxicillin, likely because offshore sites are exposed to more foreign bodies than inshore sites due to contact with more currents, so they require even more monitoring and upkeep.[8]
On top of barriers to effective treatment, there are still many unknowns with using antibiotics for treating coral diseases, such as how they affect the reef microbiome and reef inhabitants. Amoxicillin is a general antibiotic, meaning it kills all bacteria. One study found that the combination of many antibiotics, including amoxicillin, harms cyanobacteria.[10] Cyanobacteria play significant roles in sustaining rock- and coral-dwelling plant communities, make up much of the reef microbiome, contribute to calcification and are the primary nitrogen fixers for reef ecosystems.[11] However, too many cyanobacteria can lead to bacterial mats on coral surfaces, which block photosynthesis, prevent fish from reproducing in affected areas and prevent coral settlement. [12] An overabundance of cyanobacteria has also been linked to black band disease and could also lead to algae overgrowth.[13][14] So, whether amoxicillin harming cyanobacteria populations is a net positive or negative depends on the reef's health prior to amoxicillin treatment. Healthy reefs tend to have normal levels of cyanobacteria, while unhealthy reefs tend towards an overabundance of cyanobacteria.[15] More research is necessary to gain a broader sense of the effects of antibiotics, specifically amoxicillin, on reef ecosystems as a whole.
Antibiotic resistance is another concern. It is likely only a matter of time before SCTLD develops amoxicillin-resistance and another antibiotic is needed, and as of right now there are no other viable treatments for SCTLD besides amputation. The amoxicillin is also applied by hand by divers, which is regarded as "expensive, time-consuming, and limited in scale to relatively small areas", so while it is an undeniably effective method of treating SCTLD, its benefits on a larger scale may be negligible if interventions lack proper funding and resources.[16]
Potential Causes and Remedies for SCTLD
Researchers have attempted to further explore the primary causes of stony coral tissue loss disease (SCTLD) and its sources of transmission. Two possible classes of bacteria, Rhodobacterales and Rhizobiales were examined for their presence in diseased coral tissue.
To further investigate, researchers performed 16S rRNA gene sequencing on multiple coral species, including both apparently healthy and diseased Stephanocoenia intersepta (SINT), also known as blushing star coral. Researchers found signatures of the bacteria families Nitrosopumilaceae and Rhodobacteraceae in their coral samples, but were unable to relate these families with the onset of SCTLD. Both of these bacteria families are related to algal blooms, the overabundance of nitrite and nitrate, and poor water quality, so it is difficult to determine if these bacteria were already present in stressed reefs, can cause SCTLD, or if they are expressed as a result of SCTLD.
Interestingly, after sequencing the microbiome of the blushing star coral (Stephanocoenia intersepta), researchers discovered "that these [blushing star] corals may harbor disease-related bacteria without succumbing to tissue loss" and suggest it as "an interesting question for future research".[17] Lastly, the researchers also point to signatures of Rhodobacterales and Rhizobiales found in their sequencing of the surrounding water and sediment of their study in Florida as a point for future investigation. Another study that contemplated the possible sources and causes of SCTLD stated that they found "no evidence that bacteria are the primary cause of SCTLD" but that their involvement in secondary response or transmission is plausible.[18] It is possible that these bacteria classes may "co-occur, but not necessarily interact" to cause SCTLD, or that they may directly or indirectly promote the spread of SCTLD.[17]
Studying the microbiome of corals in relation to SCTLD is essential to slowing the transmission of the disease: if researchers can identify possible agents of the disease and understand their pathogenic mechanisms, scientists will be more informed about which agents to pay attention to and how to stop them.

Another aspect of Stony Coral Tissue Loss Disease is the issue of how to recolonize areas that have lost corals due to SCTLD. One such technique to recolonize is called microfragmentation, where pieces of a larger coral are cut off or 'fragmented' and then outplanted to "[take] advantage of the high regenerative ability of corals".[19] In this way, more colonies of coral are created quickly to better restore an ecosystem. However, there are reported issues with the use of microfragmentation technology, namely that rapid coral growth ceases shortly after the initial outplantation. One scientific study focused on Porites lobata, also known as lobe coral and its initial and long-term responses to microfragmentation and outplantation.
In observing lobe coral's response to fragmentation and plantation, researchers noticed a pattern of two growth cycles: "(1) rapid tissue regeneration at wound margins, followed by (2) a slower growth phase during which the colony is stabilized by new tissue deposited onto the substrate".[19] This trend is also reinforced by the graph below - rapid growth occurs in the first two weeks following fragmentation, and then a slower growth cycle takes over starting in the third week.[19] In Porites lobata, fragmentation kicked off processes such as "acute calcium homeostasis disruption and a variety of electron transport genes", which "push for wound healing" and activate the rapid growth of smaller colonies.[19] Once the site of fragmentation is closed, growth slows down a little bit and the coral returns to a state of homeostasis.
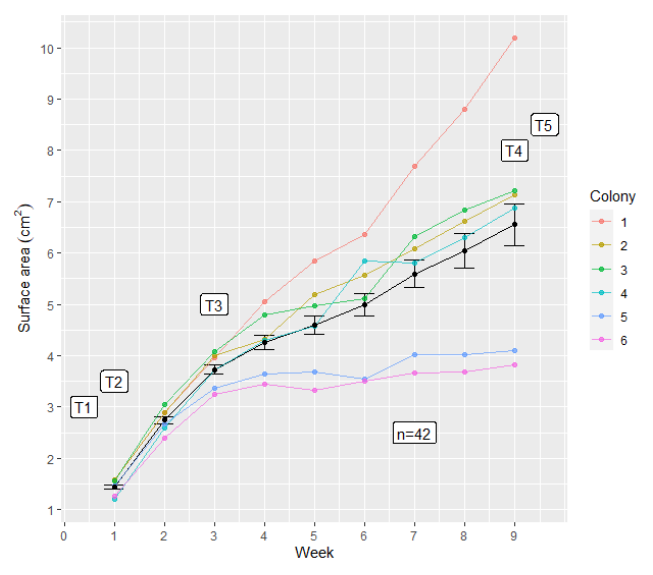
Interestingly, the process responsible for rapid coral regeneration, calcium homeostasis disruption, also leads to cell death: "sustained levels of calcium in the cytosol lead to the initiation of apoptosis".[19] Without the balancing and removal of calcium from cells, via homeostasis, cells shut themselves down and die off. Overall, the study found that "the severity and length of ER stress and calcium homeostasis disruption often determines the fate of cells in response to environmental change: survival or initiation of apoptosis".[19] This is important research to consider when choosing microfragmentation as a technique for coral reef restoration, and more needs to be done to discover the optimal conditions for fragmentation and outplantation. Additionally, if researchers investigated ways to reduce endoplasmic reticulum stress and maintain a semblance of calcium homeostasis through the rapid growth phase, this may ensure linear and consistent growth.
References
- ↑ 1.0 1.1 1.2 Traylor-Knowles N, Connelly MT, Young BD, Eaton K, Muller EM, Paul VJ, et al. Gene Expression Response to Stony Coral Tissue Loss Disease Transmission in M. cavernosa and O. faveolata From Florida [Internet]. Coral Reef Research. Frontiers in Marine Science; 2021 [cited 2022May25]. Available from: https://www.frontiersin.org/articles/10.3389/fmars.2021.681563/full
- ↑ Stony Coral Tissue Loss Disease (SCTLD) Tracking Map [Internet]. Oref.maps.arcgis.com. Atlantic and Gulf Rapid Reef Assessment; [cited 2022May25]. Available from: https://oref.maps.arcgis.com/apps/View/index.html?appid=6bf1ce3fcd8948598000aac1dda9e84a&extent=-133.8154%2C-0.5271%2C-49.4404%2C38.7200
- ↑ Study finds genes role in immune response of Florida corals to rapidly spreading disease [Internet]. Science News. ScienceDaily; 2021 [cited 2022May26]. Available from: https://www.sciencedaily.com/releases/2021/07/210706115400.htm
- ↑ Staletovich J. Research breakthrough finds hope for corals infected with stony coral disease - they can fight back [Internet]. WLRN. WLRN; 2021 [cited 2022May25]. Available from: https://www.wlrn.org/news/2021-07-12/research-breakthrough-finds-hope-for-corals-infected-with-stony-coral-disease-they-can-fight-back
- ↑ 5.0 5.1 5.2 5.3 Neely K, Shea C, Macaulay K, Hower E, Dobler M. Effectiveness of topical antibiotics in treating corals affected by Stony Coral Tissue Loss Disease. PeerJ [Internet]. 2020 [cited 26 May 2022]; pp. 1-12. Available from: https://doi.org/10.7717/peerj.9289
- ↑ Aeby G, Ushijima B, Campbell J, Jones S, Williams G, Meyer J, et al. Pathogenesis of a Tissue Loss Disease Affecting Multiple Species of Corals Along the Florida Reef Tract. Frontiers in Marine Science [Internet]. 2019 [cited 26 May 2022]; 6, 678. Available from: doi: 10.3389/fmars.2019.00678
- ↑ Miller C, May L, Moffitt Z, Woodley C. Exploratory treatments for stony coral tissue loss disease: pillar coral (Dendrogyra cylindrus). NOAA Technical Memorandum NOS NCCOS 245 and CRCP 37 [Internet]. N.d. [cited 26 May 2022]. Available from: doi:10.7289/V5/TM-NOS-NCCOS-245
- ↑ 8.0 8.1 Neely K, Shea C, Macaulay K, Hower E, Dobler M. Short- and Long-Term Effectiveness of Coral Disease Treatments. Frontiers in Marine Science [Internet]. 2021 [cited 26 May 2022]; pp. 2-19. Available from: https://core.ac.uk/outputs/481309077
- ↑ Neely, K. Coral Disease Intervention Plan. Florida DEP [Internet]. 2018 [cited 26 May 2022]; Miami, FL. P. 12.
- ↑ González-Pleiter M, Gonzalo S, Rodea-Palomares I, Leganés F, Rosal R, Boltes K et al.Toxicity of five antibiotics and their mixtures towards photosynthetic aquatic organisms: Implications for environmental risk assessment. Water Research [Internet]. 2013 [cited 26 May 2022]; 47(6) pp. 2050-2064. Available from: https://doi.org/10.1016/j.watres.2013.01.020
- ↑ Charpy L, Casareto B, Langlade M, Suzuki Y. Cyanobacteria in Coral Reef Ecosystems: A Review. Journal of Marine Sciences [Internet]. 2012 [cited 26 May 2022]. Available from: https://doi.org/10.1155/2012/259571
- ↑ Kuffner I, Walters L, Becerro M, Paul V, Ritson-Williams R, Beach K. Inhibition of coral recruitment by macroalgae and cyanobacteria. Mar Ecol-Prog Ser [Internet]. 2006 [cited 26 May 2022]; 323:107–17. Available from: https://doi.org/10.1371/journal.pone.0125445
- ↑ Carlton R, Richardson L. Oxygen and sulfide dynamics in a horizontally migrating cyanaobacterial mat—Black band disease of corals. FEMS Microbiol Ecol [Internet]. 1995 [cited 26 May 2022];18(2):155–62. Available from: doi:10.3354/meps323107
- ↑ Larned S. Nitrogen- versus phosphorus-limited growth and sources of nutrients for coral reef macroalgae. Mar Biol [Internet]. 1998 [cited 26 May 2022];132(3):409–21. Available from: https://doi.org/10.1007/s002270050407
- ↑ Brocke H, Polerecky L, de Beer D, Weber M, Claudet J, Nugues M. Organic Matter Degradation Drives Benthic Cyanobacterial Mat Abundance on Caribbean Coral Reefs. PLOS ONE [Internet]. 2015 [cited 26 May 2022]; 10(5). Available from: https://doi.org/10.1371/journal.pone.0125445
- ↑ Forrester G, Arton L, Horton A, Nickles K, Forrester L. Antibiotic Treatment Ameliorates the Impact of Stony Coral Tissue Loss Disease (SCTLD) on Coral Communities. Frontiers of Marine Science [Internet]. 2022 [cited 26 May 2022]; para. 33.
- ↑ 17.0 17.1 Rosales SM, Clark AS, Huebner LK, Ruzicka RR, Muller EM. Rhodobacterales and Rhizobiales are associated with stony coral tissue loss disease and its suspected sources of transmission [Internet]. Frontiers. Frontiers; 1AD [cited 2022May26]. Available from: https://www.frontiersin.org/articles/10.3389/fmicb.2020.00681/full#h5
- ↑ Landsberg JH, Kiryu Y, Peters EC, Wilson PW, Perry N, Waters Y, et al. Stony coral tissue loss disease in Florida is associated with disruption of host–zooxanthellae physiology [Internet]. Frontiers. Frontiers; 1AD [cited 2022May26]. Available from: https://www.frontiersin.org/articles/10.3389/fmars.2020.576013/full
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 Lock C, Bentlage B, Raymundo LJ. Calcium homeostasis disruption initiates rapid growth after micro-fragmentation in the scleractinian coral Porites lobata. bioRxiv. 2022 Jan 1.